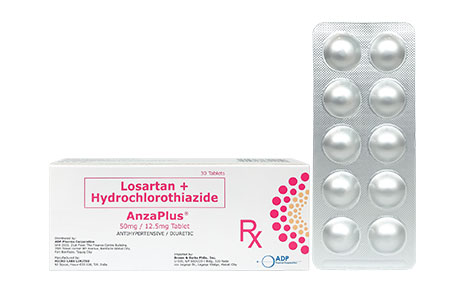
Losartan + Hydrochlorothiazide
FDA Classification
Prescription Drug (Rx)
Formulation
Each film coated tablet contains:
50mg Losartan + 12.5mg Hydrochlorothiazide
Preparation
Film Coated Tablet
Therapeutic Class
Angiotensin II Receptor Blocker/Diuretic
Physical Description
10 Tablets per Alu-Alu blister
Indication
For the management of Hypertension.
Dosage / Direction of Use
| Anzaplus 50mg/12.5mg | once a day or as prescribe by Physician |
Availability
Anzaplus 50mg/12.5mg Box of 30s/3 Blisters